 For example: Li, Na, and K are much more reactive than are Ca, Sr, and Ba Li, Na, and K form compounds with oxygen in a ratio of two of their atoms to one oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to one oxygen atom. However, the specific properties of these two groupings are notably different from each other. A second grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemical properties in common. One such grouping includes lithium (Li), sodium (Na), and potassium (K): These elements all are shiny, conduct heat and electricity well, and have similar chemical properties. Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic tableĪs early chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together by their similar chemical behaviors.Predict the general properties of elements based on their location within the periodic table.State the periodic law and explain the organization of elements in the periodic table.Sources: International Union of Pure and Applied Chemistry website, AFP, BBC, Chemistry.about. 
Their names were ratified by the IUPAC nearly a year later on element 114 became known as Flerovium (Fl) and element 116, Livermorium (Lv).īoth are highly radioactive and were the heaviest elements on the table until the recent discovery of elements 115, 117 and 118. The table's last major revision occurred in June 2011, when elements 114 and 116 were also discovered by a joint Russian-American team from Dubna's Joint Institute for Nuclear Research and California's Lawrence Livermore National Laboratory. When were new elements last added to the table? The Riken Institute had earlier said that the name Japonium (in honour of Japan) might be proposed for element 113. It is currently being used as a nuclear fuel due to its radioactivity. Thorium (Th) gained its name from Thor, the Norse God of Thunder, upon its discovery in 1828. The element Einsteinium (Es), discovered in 1952, was named in honour of renowned scientist Albert Einstein, while Europium (Eu) is named after the continent of Europe after it was isolated in 1901. After initial acceptance, the element's name and two-letter symbol are put up for public review for five months before the IUPAC Council makes its final decision. How many elements are there?Īccording to the IUPAC, new elements can be named after a mythological concept, a mineral, a place or country, a property or a scientist. It can also help predict the properties of new elements that have yet to be discovered or created.  
The table is a useful tool for people to derive relationships between the different properties of the elements. Lanthanides and actinides, often called "inner transition metals", are commonly hived off as a separate section under the main table as including all 30 - including Uranium - would make the table too wide. They are located on the left, separated from the non-metals on the right by a zig-zag line. 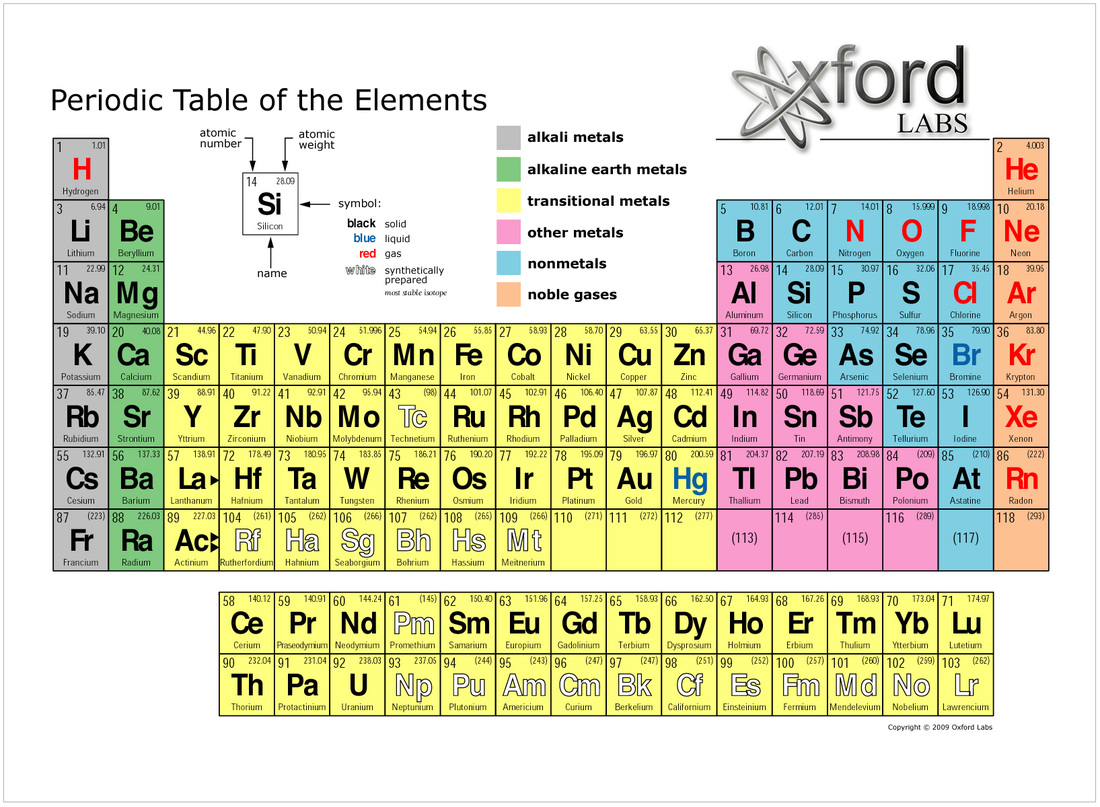
Most elements on the table are metals divided into six broad categories - alkali metals, alkaline earths, basic metals, transition metals, lanthanides and actinides. Those in the same period have the same number of atomic orbitals (the wave-like behaviour of either one or a pair of electrons in an atom) - for instance, the first period only has two elements, hydrogen and helium, with only a single orbital. There are seven rows, called periods, and 18 columns, called groups, in the table.Įlements in the same group share similar properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
